
SOLVED:BONUS QUESTION: Calculate the Gibbs energy, entropy, and enthalpy of mixing when 2.OOmol CsHja (hexane) is mixed with 3.0Omol C,H1s (heptane) at 298K; treat the solution as ideal:. (5 point) SRT V3

Gibb's free energy of mixing plus Gibb's excess energy vs. water mole... | Download Scientific Diagram

Concentration dependence of Gibbs free energy of mixing (G M /RT) and... | Download Scientific Diagram

Lecture 18Multicomponent Phase Equilibrium1 Theories of Solution The Gibbs energy of mixing is given by: And the chemical potential is: For ideal gases, - ppt download
![PDF] On the prediction of Gibbs free energy of mixing of binary liquid alloys | Semantic Scholar PDF] On the prediction of Gibbs free energy of mixing of binary liquid alloys | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/81e2e777b45d3897628323bfc42a4f91db61d2f9/5-Table1-1.png)
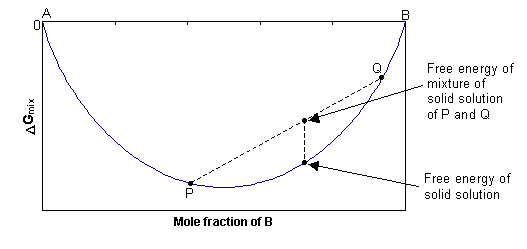
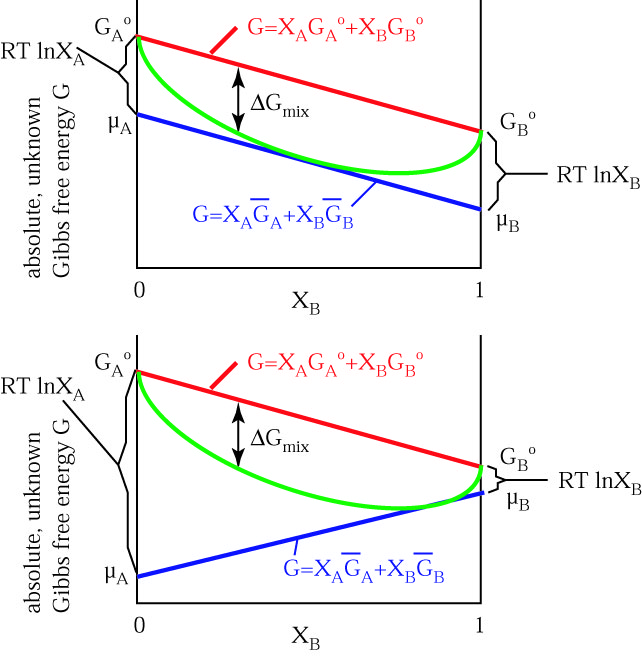
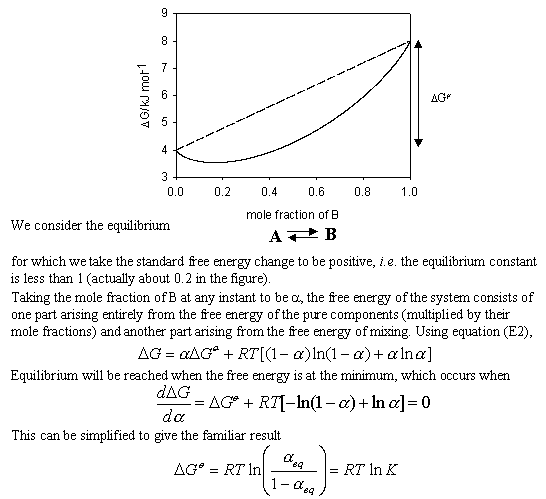

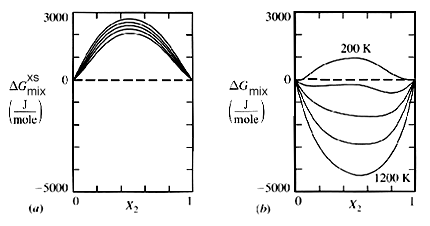
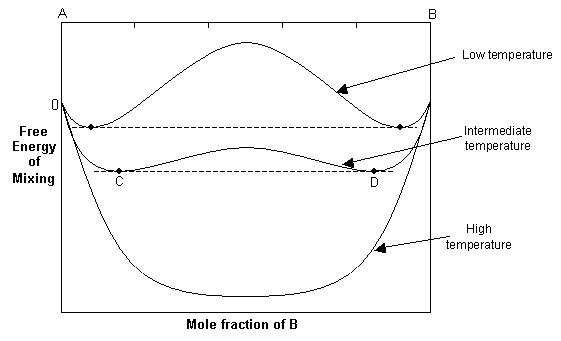

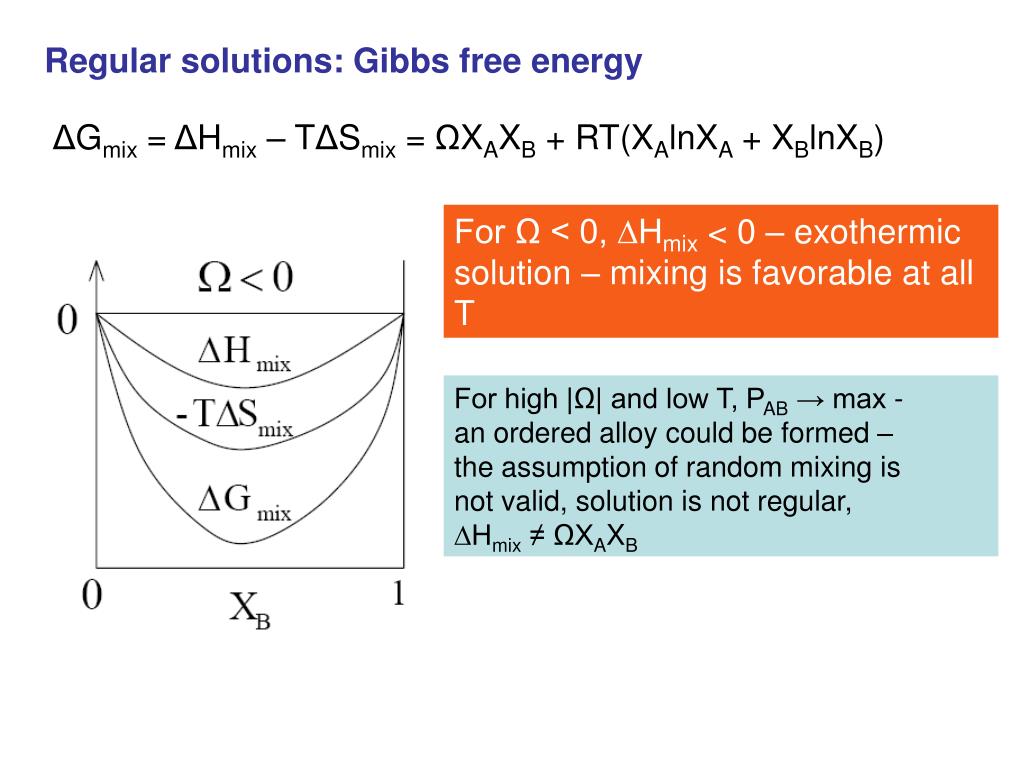



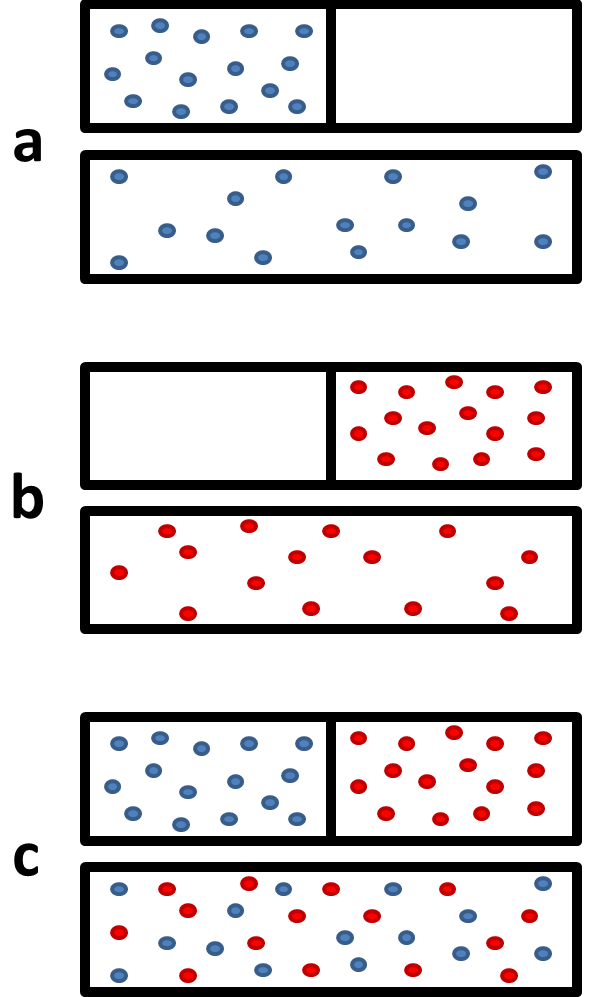

![The Gibbs energy of mixing, Delta g_mix [J/mol], in a | Chegg.com The Gibbs energy of mixing, Delta g_mix [J/mol], in a | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F471%2F47141c8f-8f8e-4905-9865-4b33d23f972b%2FphpR2OsNz.png)



